Peptidoglycan in bacterial biology
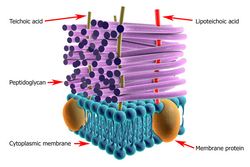
Peptidoglycan (PGN) is an essential and unique component of the bacterial cell wall. PGN metabolism plays a central role in bacterial cell structure and shape, antibiotic resistance and host-microbe interactions. PGN is the target of existing antibiotics and is also utilised by higher organisms to detect the presence of bacteria. Understanding the mechanism by which mature PGN assemble will help decipher the dynamic process of bacterial cell growth and division. The scope of the EU-funded PGNFROMSHAPETOVIR (The role of peptidoglycan in bacterial cell physiology: from bacterial shape to host-microbe interactions) project was to study the role of PGN assembly in bacterial shape. For this purpose, scientists utilised Helicobacter pylori as a model organism and aimed to develop novel therapeutic strategies to overcome the hurdle of antibiotic resistance. Their work unveiled key molecules in the machinery of PGN assembly that could be manipulated to prohibit cell wall biosynthesis. Considerable effort was devoted to the role of PGN in host-microbe interactions and the mechanism of its detection. Using several bacterial models, scientists wished to understand how pathogens subvert/modulate the host response by modifying their PGN. They observed that host detection of PGN primed the immune system to deal with the presence of commensal flora in the gastrointestinal tract. In addition, during disease, PGN substructures proved to be instrumental at restoring balance even though human pathogens devised strategies to avoid PGN recognition by the host. Taken together, the activities of the PGNFROMSHAPETOVIR project unveiled a pivotal role for PGN in stealth mechanisms, part of the virulence strategy of many human pathogens. Furthermore, the identification of PGN by the host during infection provides a novel angle on host immune responses with potential therapeutic interest.