Beating heart on a chip will speed up drug delivery
Drug development is time-consuming and expensive. The average time to develop a new single drug is 12 years, and the cost runs into billions of euro. Testing can cause unwanted side effects in human patients and animals, and many drugs also eventually fail: less than 1 % of the initially investigated candidate drugs reach the market. “The further in the drug discovery pipeline the failure occurs, the higher the waste of investment, in terms of money, time and human safety,” explains Roberta Visone, chief operating officer at BiomimX(opens in new window). The Italian SME BiomimX has created a powerful new solution in the form of a beating heart on a chip. The heart responds to drugs much as a human heart would do, letting researchers and biopharma companies run toxicity screenings on new drugs, speeding up their development. Through the EU-funded uHeart project, the company has been laying the groundwork to make the product commercially available. “These ‘beating hearts on chip’ are at this point able to exhibit human-like responses to drugs mimicking those of the native heart,” Visone notes.
Mechanical training causes heart to beat
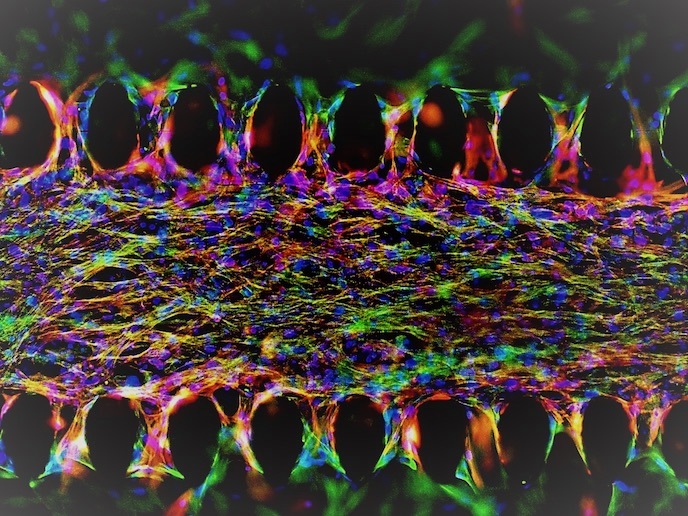
The uBeat™ patented technology provides 3D cell constructs within a controllable mechanical stimulation. This mechanical training resembles the native heartbeat and instructs the in vitro heart to beat as a natural one would. This allows researchers to accurately assess how a functional human heart would react to certain drugs, giving unprecedented levels of precision and accuracy. “uBeat™ allows us to generate predictive models of human organs and pathologies that provide invaluable insights and also to plan smarter clinical trials,” adds Visone. To grow the heart, the technology uses human induced pluripotent stem cells(opens in new window). The beating mechanics enables the generation of a human heart tissue and lifelike beating within a week. During each heartbeat, electrical impulses travel through the tissue, causing cardiac cells to contract and an extracellular matrix to mechanically deform. uHeart is also able to detect online such electrical signals thanks to a second patent-protected technology (uECG), and to monitor, in real time, electrophysiological alterations induced by drugs.
Beating success
To commercialise uHeart as a predictor of cardiotoxicity, the team is currently working on two aspects: industrial production of the device to meet the automation and throughput required by pharma companies; and pharmacological validation to prove the superior ability of uHeart in predicting cardiotoxic effect, compared to existing in vitro models and animal studies. “Both steps are ongoing thanks to active strategic collaborations,” notes Visone. The project led to confirmation that the uHeart reproducibly and accurately responds to drugs in the same way as the native heart tissue would do. “This was confirmed by testing known cardiotoxic compounds in uHeart and detecting expected functional alteration of electrophysiological activity,” says Visone. Other than proceeding with industrialisation and pharmacological validation of uHeart, the team are also focusing on its application in precision medicine. “uHeart is indeed an ideal tool for modelling specific cardiac pathologies with potentially a single patient resolution, opening the path to testing patient-specific treatments,” Visone explains. The company is also expanding their portfolio to model different organs and diseases. Mechanical stimulation is indeed essential not only to enhance cardiac tissue maturation, but also to faithfully mimic in vitro any tissue or disease state in which alteration of mechanical microenvironment exerts a pathophysiological response. Another model at the same level of development and also based on the uBeat technology, uKnee, is the first in vitro model of human joint osteoarthritis. Models of the gut, lungs, teeth and even neuromuscular circuitry are also in the pipeline.