Fibroblasts in breast cancer
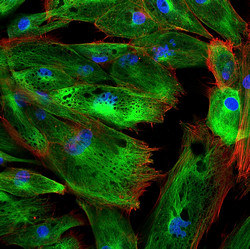
Cancer-associated fibroblasts (CAFs) are a sub-population of stromal fibroblasts that support tumour cell growth, enhance angiogenesis and help remodel the extracellular matrix (ECM). Accumulating evidence indicates that CAFs also mediate inflammation, which in breast cancer correlates with bad prognosis. The EU-funded CAF AND INFLAMMATION (Defining the molecular mechanisms of fibroblast-mediated inflammation and its role in cancer progression and metastasis) project set out to unveil the mechanism behind the role of CAFs in breast cancer. In this context, scientists found that mammary fibroblasts can be educated by breast cancer cells to become activated to a pro-inflammatory state that supports malignant progression. Through proteomic analysis, they identified a functional role for the secreted pro-inflammatory mediator osteopontin in the process. Osteopontin - implicated in inflammation, tumour progression, and metastasis - has the capacity to reprogram normal fibroblasts into tumour-promoting CAFs. Insight into the mechanism led to the discovery of a multi-protein complex involved in sensing tissue damage and promoting the secretion of inflammatory cytokines. Mortality in breast cancer is a result of metastatic disease. Understanding the mechanisms underlying the formation of a metastatic niche is therefore of fundamental importance in cancer research. Interestingly, researchers found that lung fibroblasts become activated before the formation of pulmonary metastases. This coincides with collagen deposition in the lung and pro-fibrotic signalling followed by migration of a subpopulation of lung CAFs from the bone marrow. Collectively, CAF AND INFLAMMATION study outcomes support the involvement of breast cancer CAFs in tumour maintenance and their co-evolution at the metastatic site with tumour progression. The mechanism that activates resident or migrating fibroblasts could potentially be a potent therapeutic target.