Organoids to treat liver diseases
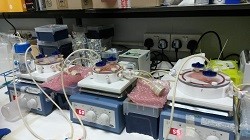
Inherited cholestatic disorders (ICDs) are characterised by defects in bile synthesis and secretion, leading to progressive liver disease. Many ICD patients do not respond to medical treatment and need liver transplantation. With 10% of patients dying while waiting on the transplant list due to shortage of liver donors, there is an imminent need for alternative therapies. Bioengineered tissues are emerging as a promising solution for reducing the need for donor organs. However, the complexity of liver organisation makes the generation of functional liver challenging. To address this issue, researchers of the ERC-funded CLOC project produced liver organoids in vitro using hepatocytes cultured on three-dimensional (3D) scaffolds. CLOC brought under its umbrella multidisciplinary experts including surgeons, engineers and cell biologists who worked together to guarantee the success of the project. “We wanted to develop novel treatments for ICDs but also models for studying liver development,″ explains Prof. Paul Gissen at the University College London, principal investigator of the CLOC project. Stem cells for liver organoids The scarce availability of viable primary hepatocytes and their poor in vivo engraftment, severely limit the success of the cell transplantation approach. On the other hand, the pluripotency of stem cells renders them an ideal source for tissue bioengineering. In particular, human induced pluripotent stem cells (hIPSCs) derived from reprogrammed skin fibroblasts by overexpression of particular factors have become a significant tool of regenerative medicine. Production of patient-specific hIPSCs can be scaled up, generating fully immuno-compatible differentiated cells for clinical application. CLOC researchers decided to seed hIPSCs onto a 3D extracellular matrix (ECM) scaffold. “We generated liver organoids from a decellularised scaffold obtained from the mouse liver. We then repopulated the scaffold with differentiating hepatocytes that were derived from hIPSCs, ″ continues Prof. Gissen. The resulting organoids preserved the micro-architecture, blood vessel network and ECM of liver. They were subsequently placed in a bioreactor where they were cultured for several weeks under a constant flow of oxygen. Compared to two-dimensional cultures, results showed that seeding hIPSCs on a 3D scaffold promoted faster cell maturation, a prerequisite for successful in vivo transplantation in an adult organism. Advantages of CLOC organoids According to Prof. Gissen, “the 3D environment provides mechanical stimuli to the cells, and alongside tissue-specific signalling, enhances liver cell function and proliferation. In addition, key ECM components assist hIPSC differentiation into hepatocytes.″ Importantly mouse liver is a readily available source of decellularised scaffolds and requires few cells to repopulate. By using hIPSCs obtained from ICD patients, CLOC researchers were able to generate in vitro models of ICDs for drug screening purposes. As Prof. Gissen outlines “the next step is to test the generated organoids in mice with liver defects and also to generate organoids from different materials such as plastic.″ Undoubtedly, the safety of transplanting hepatocytes from hIPSCs warrants further investigation. Although hepatocyte transplantation has been used to treat various metabolic disorders, direct infusion of cells will unlikely reverse polarised bile flow and correct ICDs. Liver organoids offer hope in this respect since in addition to supporting the terminal differentiation of stem cells prior to cell transplantation, they could also be used for whole organ regeneration.