Miniscule lasers within cells advance imaging in biomedical research
Following the action of cells within the body can provide answers to many biomedical questions such as cancer cell invasion and metastasis. However, existing cell labelling approaches can either only discriminate a limited number of different cells or require destructive read-out, killing the cell in the process.
Laser beads that emit at unique frequency
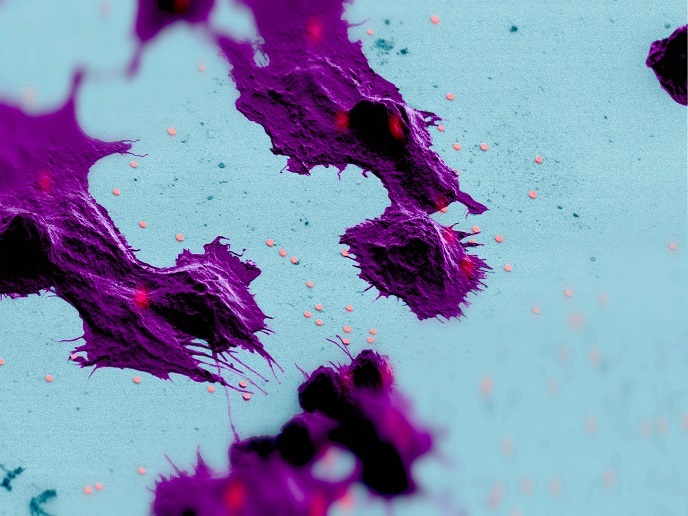
Researchers of the European Research Council (ERC)-funded ABLASE project developed tiny lasers, some with dimensions smaller than the volume of a cell nucleus, that can be efficiently inserted into cells. “Our goal was to uniquely tag cells in a way that doesn’t interfere with their properties or behaviour,″ explains ERC grantee and principal investigator Malte Gather, professor at the University of St Andrews. These micro-lasers consist of semiconductor disks or biocompatible beads with a green fluorescent dye. Upon excitation with light they emit laser light that comprises a unique composition of several narrow-band peaks depending on the size of the bead. By introducing beads of different sizes, scientists generate emission patterns that can be used to confidently distinguish each laser, much like barcodes are used to identify and track parcels. Using a molecular biology method traditionally harnessed to transfer foreign DNA into cells, ABLASE researchers have been able to incorporate the lasers into various cell types such as those involved in immune responses. This methodology enables the incorporation of laser beads into tens of thousands of cells at the same time. The team then investigated the energy these lasers require to start lasing, their size, stability and biocompatibility. For the team’s latest laser designs, lasing starts at miniscule levels of excitation light, lower than the energies biologists use in many modern optical microscopes, which minimises the impact on the cells. The cellular lasers also demonstrated the ability to remain stable and within cells during various cellular processes. Intriguingly, cells transfer the lasers to their progeny without any obvious impact on key cell functions, facilitating cell tracking for many generations. By inserting multiple lasers into the mother cells, the ABLASE approach can follow all progenies of a cell and also uniquely label more cells than previously thought.
Research significance and future prospects
ABLASE project activities have advanced the use of single cell lasers in biomedical research with the prospect of gaining insight into fundamental biological processes such as how cells migrate within the body. “The use of single cell lasers is a novel concept, and so naturally ABLASE has been concerned with proof-of-concept work, basic research and development, as well as with first steps towards applications,” emphasises Gather. Future plans include further miniaturisation of the lasers to make their insertion into cells even easier, and a higher-throughput laser fabrication process. Furthermore, ABLASE researchers hope to test the lasers in vivo and extend their applications enhancing their performance and make them respond to changes in biological markers inside the cell. Undoubtedly, single cell lasers require a number of further technological improvements to be fully applicable. Nonetheless they have immense potential in cell tagging applications and can be used for sensing within living cells and tissues. Since the beginning of ABLASE, other research groups around the world have made significant contributions to the emerging field of single cell lasers, reinforcing its development potential.