Solving the problem of temperature-sensitive vaccines
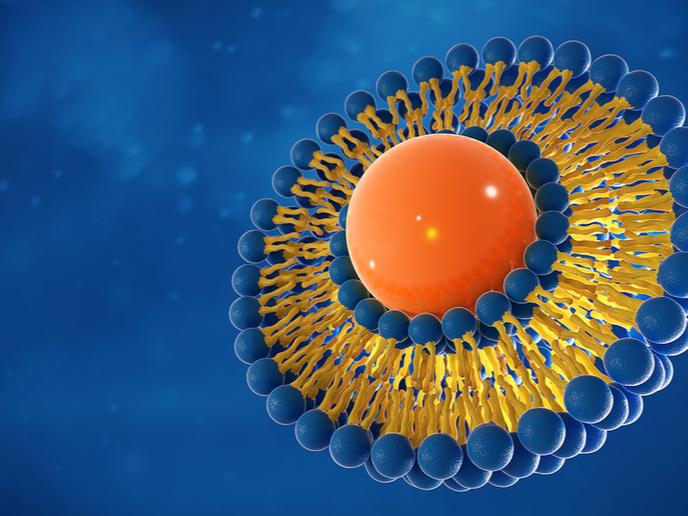
When vaccines are exposed to temperatures outside the recommended ranges, they can lose their potency and effectiveness. Ensuring that vaccines are properly transported and stored is therefore an important step in successfully immunising people against vaccine-preventable diseases. However, this becomes challenging when handling vaccines that need to be kept at below-freezing temperatures. Researchers from Graz University of Technology (TU Graz) in Austria and The University of Texas at Dallas (UT Dallas) in the United States have now demonstrated a new technique that could help solve this problem. The low-cost method was developed with partial support from the EU-funded POPCRYSTAL project, coordinated by TU Graz. As described in the study(opens in new window) published in the journal ‘Nature Communications’, it involves creating crystalline exoskeletons around delicate liposomes and other lipid nanoparticles, and then stabilising them at room temperature for up to two months. Liposomes and lipid nanoparticles are both excellent drug delivery vehicles that transport the drug inside a protective outer layer of lipids. However, they’re also both thermodynamically unstable at room temperature. Two COVID-19 vaccines that use lipid nanoparticles are the Moderna and Pfizer/BioNTech vaccines. “The expense of keeping these vaccines very cold from the time they’re made to the time they’re delivered is a challenge that needs to be addressed, especially because many countries don’t have sufficient infrastructure to maintain this kind of cold chain,” stated study co-author Dr Jeremiah Gassensmith of UT Dallas in an article(opens in new window) posted on the university’s website. “Although we did not include in this work the specific lipid nanoparticles used in current COVID-19 vaccines, our findings are a step toward stabilizing a lipid nanoparticle in a way that’s never been done before, so far as we know.” Membrane proteins are found in lipid bilayers, which are cell membranes that have two layers of lipid molecules. According to co-author Gabriele Meloni, also of UT Dallas, one of the research challenges “is that both membrane proteins and lipid bilayers are very delicate and intrinsically metastable, and we’re trying to combine them in order to understand how these proteins function.” They also require careful handling and need to be prepared fresh every time. “They cannot be stored for long periods and are not easily shipped to colleagues in other labs,” Meloni added.
Stabilising the system
The research team developed a technique to stabilise this lipid system and demonstrated the results using transmembrane proteins – a type of membrane protein that spans the entire cell membrane. They mixed liposomes with two chemicals (zinc acetate dihydrate and methylimidazole) in a buffer solution, resulting in the formation of a crystal matrix around each liposome. “We think that the lipids interact with the zinc just strongly enough to form an initial zinc-methylimidazole structure that then grows around the lipid sphere and completely envelops it, like an exoskeleton,” observed Dr Gassensmith. “We sort of co-opted nature in creating this totally fake shell, where the biomacromolecules — the lipids and proteins — catalyze the growth of this exoskeleton.” To release and reconstitute the liposomes, the team used ethylenediaminetetraacetic acid, a potent zinc-chelating agent widely used in medicine and industry because it can chelate(opens in new window) metals to form stable, water-soluble complexes. POPCRYSTAL (Precisely Oriented Porous Crystalline Films and Patterns) aims to create novel crystalline metal-organic framework films and patterns with precisely oriented nanopores and nanochannels. For more information, please see: POPCRYSTAL project(opens in new window)