Non-destructive 3D imaging to help mend broken hearts
Currently, the diagnosis of cardiovascular disease (CVD) relies on assessing cardiac macro-anatomy, whole heart function, and whole-body metabolic markers. This leads to 20 % of cases being misdiagnosed, and in elderly patients up to 60 % of CVDs remaining undiagnosed.
Studying heart micro-anatomy and metabolism
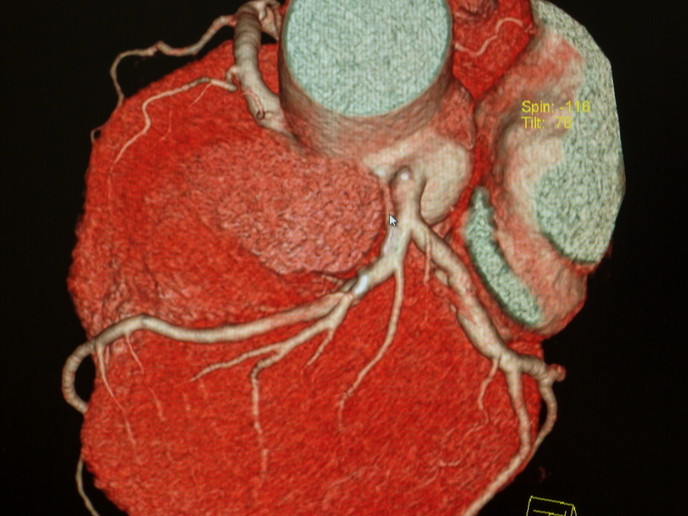
With the support of the Marie Skłodowska-Curie programme, the ANATOMY-FOUND project proposed to study the events in CVD development that occur before the macro-pathologies – routinely used for diagnosis – can be detected. For this, clinicians require organ-specific non-invasive imaging techniques to examine the micro-anatomy and metabolism of the heart, which can then be correlated with cardiac function. “Early CVD processes often begin in the heart cells, before evolving to organ-wide disease, thus new technologies that provide high-resolution 3D micro-anatomy, and organ-specific regional metabolic assessment are required,″ explains Marie Skłodowska-Curie fellow Robert Stanley Stephenson. To investigate the micro-anatomy of the heart, researchers employed contrast-enhanced micro-computed tomography (micro-CT), an ex vivo X-ray-based technique that uses an iodine-based contrast agent. This helps distinguish fat, muscle, conduction system and connective tissue at spatial resolutions approaching the single cell level. Using this approach, researchers observed the cardiac conduction system in 3D in normal and congenitally malformed human hearts for the first time. In addition, they studied the arrangement of the myocyte chains using micro-CT and diffusion tensor magnetic resonance imaging (DT-MRI), and demonstrated that myocyte orientation is modified in diseased hearts. The scan data showed that the major contributor to myocardial deformation, and thus pump-function, is not changes in myocyte chain orientation, but the rearrangement of the aggregated units of myocytes. A new in vivo metabolic imaging technique using radiolabelled pyruvate as an imaging probe, showed a reduced aerobic metabolic potential with concomitant shift to an undesirable anaerobic metabolic state in failing hearts. Interestingly, this alteration correlated with gross morphological remodelling.
ANATOMY-FOUND impact
Via novel state-of-the-art non-destructive 3D and 4D imaging methodologies, ANATOMY-FOUND successfully investigated the micro-structure and function of the heart in health and disease. The results provide a more precise understanding of the cardiac conduction system and how it is remodelled in congenitally malformed hearts. Given that cardiac conduction is the biological pacemaker of the heart, it is important that the clinicians do not damage these cells during surgery or interventions. By providing a precise 3D model of cardiac conduction, ANATOMY-FOUND hopes to make surgery safer. Towards this goal, project partners have produced the first 3D-printed hearts fabricated with transparent flexible myocardium to illustrate a coloured cardiac conduction system. This approach will improve surgical planning and hopefully reduce mortality and morbidity in patients with congenitally malformed hearts. Project findings suggest that metabolic impairment is associated with cardiac remodelling and can manifest as pump dysfunction and heart failure. The in vivo metabolic imaging technique used in ANATOMY-FOUND has great potential as a tool for prompt CVD diagnosis, and could well become an everyday screening technique in the clinics. As researcher Michael Pedersen emphasises: “ANATOMY-FOUND provides a potential new approach for diagnosing heart disease before using traditional morphological-based methods.″ Its ability to identify regional changes in the heart could also pave the way for targeted treatments in the future.