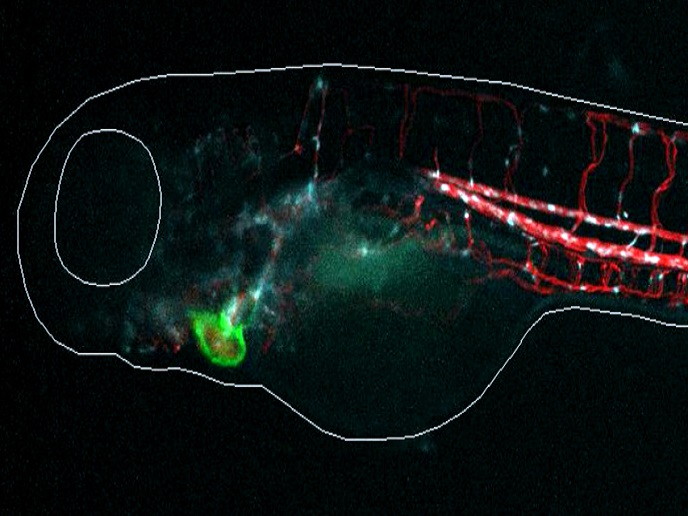
Zebrafish cardiovascular system to identify and test cardiotoxic and cardiotherapeutic drugs
High-throughput screening (HTS) using in vitro cell-based assays was once thought to be the answer to the ever-increasing flow of new potential therapeutics. However, evidence shows this method to be incomplete for understanding drug candidate performance.
HTS in zebrafish reduces later use of animals in preclinical trials
The answer, developed by the ZECARDIO(opens in new window) project, is a revolutionary HTS platform, ZeCardio®. “Originally designed for addressing drug-induced cardiotoxicity, after testing, we realised that we could also use the platform for discovering novel therapeutic drugs and perform target validation studies with the use of CRISPR technology,” outlines Dr Javier Terriente, chief scientific officer of ZeClinics, the project coordinator. ZECARDIO researchers tested and validated the platform in a preclinical setting using 100 compounds with known cardiotoxic/cardioactive profiles in humans. According to Dr Terriente, “Our results, recently published in Toxicological Sciences(opens in new window), demonstrate that zebrafish is a much more predictive experimental model than cellular systems and mice, and on average with dogs – the gold standard animal model in preclinical drug discovery in the cardiovascular area.”
Size and profitability of research increasing in leaps and bounds
Funds obtained through the EU, under ‘Supporting Innovative SMEs in the healthcare biotechnology sector’, have helped enormously with expansion of the company. “We now have close to 30 employees and growing,” enthuses Dr Terriente. Business success has been so overwhelming that ZeClinics has launched a new spin-off company, www.zecardiotherapeutics.com (ZeCardio Therapeutics). “We are currently in a series A round looking for EUR 5 million to take the different therapeutic leads we have discovered through the ZeCardio® platform towards clinical phases,” the coordinator notes. Challenges mitigated by research team working together, the key to success! Implementation of the hardware in the ZeCardio® screening platform took several more months than originally planned. “Thanks to the talent and commitment of the ZeClinics scientific team, headed by Dr Sylvia Dyballa, these problems were solved,” explains Dr Terriente. Other challenges were more administrative. “We were also very lucky to have a very talented grant manager, Vaclovas Radvilas, who helped us respond to the dynamic needs of the project,” he adds. “Our to-do list wouldn’t fit in this article,” Dr Terriente comments wryly, but summarises this in four succinct points. To consolidate the position of ZeClinics as the largest and most innovative company providing services based on the use of zebrafish obviously comes first. Second, to secure funding for ZeCardio Therapeutics and advance our drug discovery pipelines. Implementation of the GLP certification in our facilities and, lastly, to contribute further towards the industrial and regulatory acceptance of the zebrafish model for discovering new drugs. “We aim to reach this goal sooner than later as the project is part of multicentre initiatives such as the National Institutes of Health (NIH) in the United States.” Summarising, Dr Terriente points out that the SME instrument award gave ZECARDIO funds, but most importantly, it gave them strong confidence in the company they founded 6 years ago. “But the most valuable point is that the EU funding has allowed us to consolidate a highly skilled and committed team. Without them, we certainly wouldn’t be where we are now!”