Induction and transmission of genomic instability after irradiation
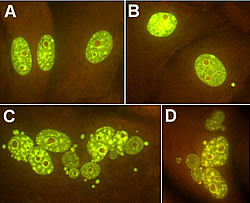
After exposure to ionising radiation, the energy deposit on the cell nucleus and the potential DNA effect play an important role in the biological damages caused. Thereby, during first cell division, radiation induced changes take place, while cell clone carrying mutations in specific genes is responsible for the damaging effects. Despite these already known facts, recent research revealed that indirect effects may also take place at very low doses, such as genomic instability and bystander effects in non-radiated cells. Due to the potential uncertainties in dosimetry carried out by epidemiological studies lower risk estimates may be assessed. These have already led to increased controversy over the adequacy of radiation protection measures for both public and radiation workers. One the other hand, biological models are able to provide data on both direct radiation effects and non-targeted effects that play a key role at the low dose region. This is why this project adopted cellular and animal models instead of conventional radiobiology studies to investigate the effect of very low doses. The RADINSTAB project examined in detail two mechanisms that greatly contribute to the cellular response. Firstly, the bystander effect that signals damage caused by targeted cells to untargeted cells has been demonstrated that at low doses induces and preserves genomic instability. Secondly, the low-dose Hyper-Radio Sensitivity (HRS) and Increased Radio Resistance (IRR) that may constitute an integrated index of the change in the dose affect relationship, around 0,5-1 Gy of low Linear Energy Transfer (LET) radiation. The interesting results derived showed that there is a considerable variation in expression of both mechanisms, while the impact of very low doses is larger than expected. Cell lines with a large bystander effect do not show induced radio resistance and vice versa. In addition, cell lines with a radiation dose response curve with a wide shoulder may not present any HRS/IRR and bystander effects. Nevertheless, research on the genetic basis of this variability is ongoing. The picture shows images of normal porcine urothelial cells within the explant outgrowth (A), bystander damaged micronucleated cells (B) and apoptotic cells (C, D) stained with acridine orange (from the paper: Belyakov, O.V. Folkard, M., Mothersill, C., Prise, K.M. and Michael, B.D. (2003) A proliferation-dependent bystander effect in primary porcine and human urothelial explants in response to targeted irradiation. Br J Cancer, 88:5, 767-74).