Budding yeast to study Gap1 functioning
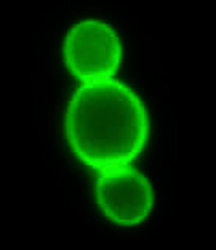
Research into yeast nutrient sensing and signalling is a long-term goal at KULeuven in Belgium as it is important from both industrial and biomedical points of view. The Yeast sensing project has so far concentrated on studying general amino acid permease (Gap1) functioning from Saccharomyces cerevisiae (budding yeast). This protein is called a transceptor as it has both transporter and receptor functions. Gap1 is also an amino acid transporter that functions as an amino acid sensor, too, for activation of the protein kinase A (PKA) pathway in yeast. Researchers are investigating the relationship between Gap1-mediated amino acid activation of the PKA pathway, and focusing on highlighting the components in the signalling pathway between Gap1 and PKA. Analysing these mechanisms will offer new insights into how the nutrient signal is transmitted from nutrient transceptor to the central growth regulator pathway controlled by PKA. The main achievements to date include increasing trehalase activity from about one hour to a window of three hours. In addition, this is the first time yeast has been used where conditions are shown to enable a transceptor to continue signalling after being endocytosed. One unexpected result reveals the ability of the Gap1 transceptor to take up amino acids as well as specific types of dipeptides. Yeast sensing will continue experiments at the molecular level to gain a better understanding of how intracellular signalling actually takes place, and also how it is stopped under normal circumstances. Additional research is also being conducted to discover whether Npr1 is a substrate for PKA. This is important since Npr1 kinase activity is needed to stabilise Gap1 and other nutrient transporters at the plasma membrane.