Helping NETs to catch bacteria in the bloodstream
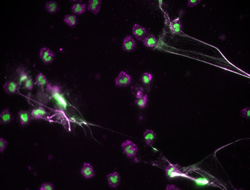
Innate immune responses to bacterial infection include phagocytosis where white blood cells ingest roaming bacteria. Another cunning mechanism has recently been identified – activated neutrophils (a type of white cell) release neutrophil extracellular traps (NETs). As the name implies, these consist of web-like fibres of DNA decorated with anti-microbial proteins that physically trap and kill microbes. Patient outcome is poorer when there are higher levels of free circulating plasma DNA in sepsis. NETs are undoubtedly a crucial part of fighting bacteria in infectious disease and the Netssepsis project aimed to investigate the molecular mechanisms that regulate the formation of NETs. Project scientists followed the trail of an enzyme, 5-lipoxygenase (5-LOX) known to be important in the regulation of inflammation. Using state-of-the-art lipidomic techniques, the researchers discovered a family of lipids formed by 5-LOX in the membranes of neutrophils in response to bacteria. Elevated levels of these 5-LOX derived lipids were also found in the peritoneal waste in dialysis patients and in mice with peritonitis and sepsis, consistent with response to infection. In the laboratory, formation of NETs was significantly reduced by addition of these neutrophil 5-LOX products. One problem with NETs is that although they are important in anti-microbial defence, they are formed at the expense of tissue injury. The novel 5-LOX products may be used to regulate neutrophil function in peritonitis and sepsis to avoid tissue damage. Future research into exactly how 5-LOX derived lipids modulate the action of the immune system may lead to the design of new anti-inflammatory therapies. Mimicking or preventing action of the new molecules could form the basis of a whole new repertoire of regulators. Patients at high risk of peritonitis such as those on dialysis or following abdominal surgery could benefit from this medical breakthrough.