Dissecting the neuromuscular junction
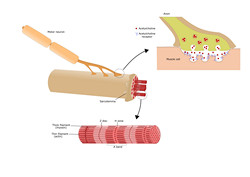
Communication between neurons and muscle takes place at the neuromuscular junction, a specific synapse where sequential molecular and biochemical events culminate in muscle tension. Although the molecular mechanisms for neuromuscular junction formation and maintenance are implicated in many pathological events, a full understanding of these mechanisms is still lacking. The scope of the EU-funded RETROGRADE SIGNALING (Molecular mechanisms of neurodegeneration) project was to elucidate the essential communication mechanisms for cell survival and synapse maintenance between neurons and their environment. Since neuronal function depends on cell-cell communication mediated by ligand-receptor interaction, the aim was to define the implicated molecules. The consortium built a microfluidic platform that recapitulates neuromuscular junctions. The system contains motor neuron cell bodies in one compartment, and muscle cells in the other, connected via motor axons that form functional neuromuscular junctions. Using this platform, researchers demonstrated that the glial cell-derived neurotrophic factor functions in a spatially specific manner to facilitate axon growth and innervation. In another part of the project, they investigated the mechanism by which the rabies virus reaches the central nervous system. Using live cell imaging, they discovered that the virus binds to the p75 neurotrophin receptor and enters the retrograde axonal transport machinery. In addition, the virus seems to exert a positive influence on the kinetics of the transport, thus facilitating its own arrival at the central nervous system. Overall, this study provided important information on the spatiotemporal distribution of ligands and receptors and unveiled the nature of the signals that are essential to neuronal health. Since the basic mechanisms of neurodegenerative diseases are poorly understood, the results of this project may open up new avenues for future drug development.