Medical device more accurately analyses airflow in the nasal cavity
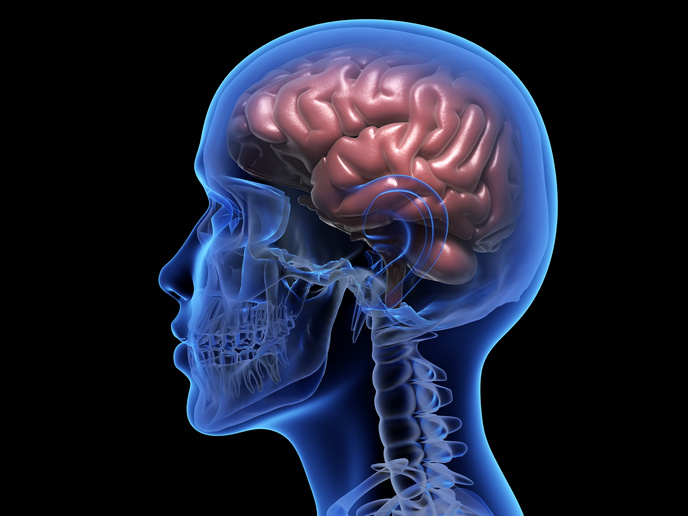
Nasal airway obstruction (NAO) is when a patient is unable to get enough air into their upper airways to be able to breath quietly, without the perception that breathing is obstructed. It is one of the most common ear, nose and throat (ENT) complaints in primary care and one of the most common reasons for secondary care referrals for ENT. It is thought that over 30 % of the world’s population suffer from NAO(opens in new window). The EU-funded NASALFLOW project has developed a diagnostic service for the healthcare market and has successfully passed clinical trials to receive its CE marking(opens in new window). EU support helped the project further assess the technical, commercial and financial viability of their solution prior to being released into the market.
Interactive visualisation and simulation
NAO is usually caused by anatomic factors. Symptoms often include headaches, shortness of breath, fatigue, sleep disturbance and a general decline in quality of life. Diagnosis is difficult as there is a lack of a reliable and objective means to measure symptoms. Current methods, including active anterior rhinomanometry (RM), peak nasal inspiratory flow (PNIF) and acoustic rhinometry (AR), are invasive and not detailed enough for clinical NAO practice. According to a recent study(opens in new window), the majority of ENT surgeons diagnose nasal obstruction using only the patient’s clinical records and nasal endoscopy. This means that, currently, the decision to proceed with surgery is mainly based on a surgeon’s experience and intuition. Reported failure rates as high as 25 % to 50 %(opens in new window). NASALFLOW’s process starts with a 3D reconstruction of the patient’s upper airway, created after the air cavity images have been isolated from an anonymised patient CT scan of the nose area. Computer modelling then finds the fluid flow solution for every point inside the nasal cavity, including data about velocity, pressure, shear stresses and temperature. The analysis, which takes under 2 hours, can then be used to support clinical decision-making, as it is complemented with the Virtual Surgery (VS) tool. This enables surgeons to simulate the likely outcomes of different interventions. A sensitivity and specificity study was conducted for the project’s NAO diagnosis at the Gregorio Marañón Hospital, Madrid(opens in new window) in 2018-2019. The results show that the technique achieved the high values of 0.85 for sensitivity (ability to identify those with illness) and 0.79 specificity (ability to identify those without illness). The results of each stage were also verified by performing 200 simulations in 100 patients.
The gold standard
Currently, to get the technology closer to market, the team is working on improving the device’s diagnostics, which will require clinical validation. The goal is to exploit the full potential of computational fluid dynamics (CFD) technology for human respiration. It could be used to diagnose different respiratory conditions relevant to paediatrics, sports or geriatrics. It can also simulate the transportation and deposition of particles in the nasal cavity. This would increase the basic understanding of the processes behind the condition and validate the efficacy of nasal sprays. “Our aim is to become the gold standard in NAO diagnosis and decision-making among ENT specialists. Our Virtual Surgery tool can avoid unnecessary surgery along with reducing failure rates and related medical expenses. Currently, there is no company more advanced than us in applying CFD to human nasal airways,” says Guillermo Sanjuán, project coordinator.