A urine test for bladder cancer diagnosis
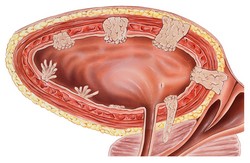
Bladder cancer is characterised by a high recurrence rate and the highest per patient treatment costs, posing a significant healthcare burden. Currently, clinical diagnosis and monitoring relies on invasive cystoscopy, emphasising the need for non-invasive and cost-effective solutions such as the identification of urine biomarkers. The scope of the EU-funded TRANSBIOBC(opens in new window) (Translation of novel biomarkers for bladder cancer for clinical outcome prediction) project was to develop an in vitro diagnostic test for monitoring bladder cancer recurrence and progression in routine clinical practice. Researchers advanced on powerful technological platforms such as capillary electrophoresis mass spectrometry (CE-MS) and micro-capillary ELISA. Following validation of the analytical performance of these platforms and assays, scientists went on to analyse well-characterised clinical samples from existing bladder cancer cohorts. Through the development of database platforms, they correlated biomarker levels with clinical information. Biomarker profiling by CE-MS led to the identification of proteins present in the urine of bladder cancer patients such as profilin-1 and histone 2B. However, the majority of the biomarkers could not be detected via ELISA due to assay interference by the urine sample. Therefore, researchers developed mass spectrometry-based assays for the quantification of these bladder cancer protein biomarkers, which exhibited superior detection performance compared to ELISA. Overall, the consortium analysed over 1 400 samples from patients across Europe by CE-MS. The peptide biomarkers provided complementary information, outperforming cytology, and were further utilised for primary diagnosis and detection of tumour relapse. This list of biomarkers was further validated in a Phase II trial and the work culminated with a prototype kit for the CE-MS-based diagnosis of bladder cancer. Considering the numerous ongoing trials of bladder cancer interventions, the TRANSBIOBC in vitro diagnostic kit, offers a fast and effective evaluation of the outcome of these therapeutic agents. Partners envisage not only early detection of recurrent BC, but also rapid and cost-effective prediction of tumour progression.