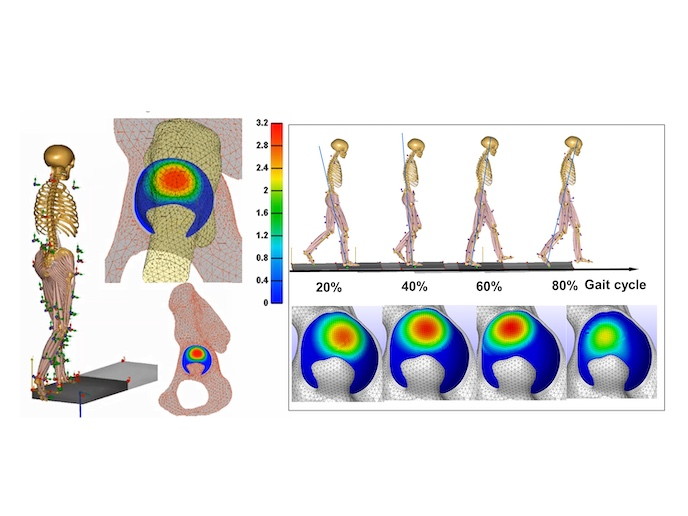
Simulating dynamic joint loading for next generation osteoarthritis treatments
Osteoarthritis (OA), where the sufferer’s joints become painful and stiff, is debilitating, reducing mobility and the ability to perform even everyday activities. Current treatments are limited to pain management, short-term relief by steroid or hyaluronan injection and strength therapy. For severe OA, options are cell or tissue transplants (with limited success), or joint arthroplasty. While joint arthroplasty is generally successful, early failure remains a concern; usually due to implant wear and loosening, necessitating surgical correction. With EU-funding the MSCHIPBIO project combined advanced computer simulation methods to link detailed predictions of hip joint loading at the whole-body scale, with joint-scale simulations of cartilage response to loading for a variety of daily activities. These dynamic loading profiles, developed at ETH Zurich(opens in new window), could help make the next-generation of synthetic implants more accurately mimic natural performance, and even surpass it.
Building biomechanical profiles
Two of the most promising methods for alleviating severe OA are tissue sparing and regeneration treatments. In tissue sparing, either the natural joint is surgically reshaped to return the joint closer to its natural form and function, or small tissue (e.g. cartilage) from donor sites on the body that don’t carry loads is transplanted to the damaged areas. Regenerative treatments transplant materials that are potentially more biologically active than the patient’s own cartilage (which succumbs to age-related degeneration). Promising candidates are soft biomaterials mimicking natural cartilage, supplemented with biological growth factors and/or donor stem cells which stimulate the growth of new cartilage tissue. However, the challenge remains of finding a material strong enough to withstand interim loading while the new tissue grows into the injury site. “One option is to use MSCHIPBIO’s models to study the potential outcome of tissue sparing treatments, so we can optimise repositioning or reshaping to bring internal joint stresses back to normal levels,” says Dr Xijin Hua, lead researcher. “The models could also provide guides for prosthetic joints to mimic the behaviour of natural self-lubricating joints and so increase their longevity.” One unexpected outcome of the simulations was evidence of a ‘self-sealing’ function in the natural hip joint, where extended periods of loading lead to a decrease in the stress within the cartilage at the edges of the joint, potentially decreasing the risk of tissue damage.
Modelling work wanted
The body-scale models are currently available within a repository of the AnyBody Modelling System(opens in new window), the simulation framework used for their creation, and are available to all this software’s licence users. The design of the joint-scale model – based on the free, public-access simulation software FEBio(opens in new window) – will be outlined in sufficient detail in a forthcoming publication for reproduction by other biomedical researchers. There is interest from orthopaedic implant manufacturers in using such models to replace some of their pre-clinical laboratory testing, enabling more robust implants to be made faster. The models could also eventually be introduced into general surgical planning software packages. “Collectively these options could influence the future treatment of OA, hopefully leading to improved care and reduced healthcare costs,” says Professor Stephen Ferguson, research supervisor. Dr Hua plans to continue the integration of the different model scales to explore a broader range of loading conditions for the human hip joint, and to also examine the performance of prosthetic joints or cartilage transplantation therapies. This research was undertaken with the support of the Marie Skłodowska-Curie programme.