The colonoscopy magic eye: novel software detects missed lesions
Traditionally, colon cancer screening takes place through colonoscopy. Colonoscopy screening depends on individual anatomy and the quality of bowel preparation. Despite technological advancements such us optics or endoscope attachments to improve visualisation and resolution, there are still procedural limitations, which may leave up to 20 % of polyps undetected. This leads to more than 44 500 cases of preventable cancer in Europe every year.
Bringing AI into colonoscopy screening
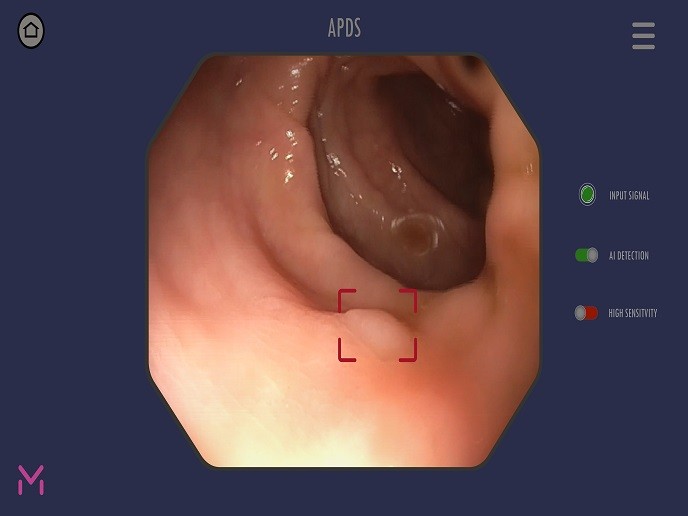
To address this limitation, the EU-funded Magentiq Eye LTD(opens in new window) project has developed an automatic polyp detection system (APDS) that points out suspicious lesions in real-time for further inspection and removal. APDS is an AI system that uses deep learning and computer vision techniques to assist doctors in the colonoscopy examination test. “APDS works as an additional eye, highlighting any polyps or lesions that appear on the physician’s screen during the examination,″ explains project coordinator and Magentiq Eye LTD CEO Dror Zur. The product comprises an add-on device that acquires digital video output signal from the endoscopic camera and runs a deep learning and computer vision algorithm in real time, similar to face or pattern recognition. The system alerts the doctor on suspicious areas by highlighting them on the main video screen. The doctor can quickly zoom in to take a closer look and decide if indeed removal of the tissue is required. Moreover, APDS has the capacity to run in offline mode on pre-recorded colonoscopy videos and scan each one for missed polyps. The system provides a video containing the areas where suspicious tissue is detected and highlighted, thereby potentially reducing the polyp miss rate, and hence, the occurrence of cancer. Preliminary data on APDS offline system performance indicate 90 % sensitivity and over 98 % specificity of lesion detection. Compared to standard colonoscopy examination, the real-time APDS system is estimated to increase the polyp detection rate by 9 % and the adenoma detection rate by 3 %. Importantly, the diagnostic capabilities of algorithms improve as they are exposed to more relevant data, in the same way as the human physician with experience.
Future prospects
During MagentiqEye, partners sought funding to conduct a feasibility study to prove technical and commercial viability. They conducted an analytical exercise to understand the market, identify potential clients and define the future commercialisation and distribution processes. Engagement with key opinion leaders from Europe and the United States (US) enabled partners to position the product with respect to end-user requirements, improve the company’s business plan, and produce a very detailed pricing sensitivity analysis. Collectively, these achievements helped towards the identification of additional technology features needed to support the APDS system and laid the foundation towards clinical trials. So far, three medical centres in Israel, Europe and the US have confirmed their participation in a real-time APDS system clinical trial that will commence in 2020 following FDA clearance. In view of the future, Zur is confident that “ongoing APDS technology development will further advance product performance and bring AI into routine colonoscopy, minimising errors.″ Long-term this will increase adenoma detection rate and contribute towards colorectal cancer prevention.