A novel bispecific antibody therapy against colorectal cancer
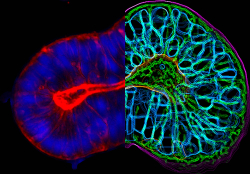
Traditionally, most cancer drugs have been based on agents that block essential functions and kill dividing cells often resulting in harmful side-effects that diminish patient quality of life. Recent drug development has focused on targeted therapies that have less toxicity. Among these, antibody-based therapy for cancer has become an established strategy for treating patients with different kinds of tumours. Human antibodies engineered to bind specifically to molecular targets present on the surface of cancer cells are indistinguishable from the natural antibodies the body makes to fight viral and bacterial infections. They retain the Y-shape of natural antibodies, and the variable antigen-binding region of the molecule has been designed to bind specific epitopes of cancer cell surface molecules. Scientists on the EU-funded SUPPRESSTEM(opens in new window) (Testing and validation of bispecific antibody combinations targeting treatment-resistant cancer stem cells using organoid-based screening tools: a new drug discovery paradigm) project exploited this technology to produce human antibodies to treat CRC. Drug development SUPPRESSTEM researchers based their strategy on research showing that resistance to current therapies can be attributed to the presence of a small subpopulation of cancer stem cells that remains after treatment and is responsible for the re-establishment of tumours and eventual disease relapse. As explained by the consortium, 'the innovation behind the SUPPRESSTEM approach was the development of therapeutics that specifically targeted cancer stem cells in CRC in order to shut down important growth and differentiation pathways'. This was achieved through bispecific antibodies that contained two different antigen-binding sites and had the capacity to bind to two different antigens or epitopes. The SUPPRESSTEM therapeutic antibodies consisted of an EGFR-targeting arm combined with an arm against LGR5, a surface molecule implicated in the Wnt signalling pathway, the most commonly altered pathway in CRC. EGFR is one of the best described survival signals in cancer, while LGR5 was originally found to mark intestinal stem cells and cancer stem cells of various types. As a result, binding and blocking these oncogenic and proliferative signals should help eliminate CRC stem cells and demonstrate an overall antitumour effect. Drug screening in organoids The high failure rate in cancer drug research has been linked to the poor predictive capacity of drug screening models for cancer and the inability to select the right patients for treatment. 'Our drug was screened on a large panel of patient material and its effectiveness was linked to a particular genetic profile', notes project coordinator Dr Mark Throsby. Towards this goal, researchers developed a patient-derived organoid assay in collaboration with leading hospitals across Europe. Recently, the organoid technology has been recognised as a novel paradigm technology capable of modelling human disease. 'The organoid technology is suitable for both preclinical drug development and for clinical purposes to predict personalised treatments', Dr Hans Clevers of the Hubrecht Institute and inventor of the Organoid technology explains. The technology is being explored to determine treatment outcome of patients in clinical trials and to identify new patient groups that benefit from specific therapies. The next goal of the SUPPRESSTEM consortium is to commercialise the patient-derived organoid assay and establish it as a universal clinical tool. Regarding the bispecific antibodies, further development for clinical testing is necessary to conclusively determine their antitumour potential in CRC patients.