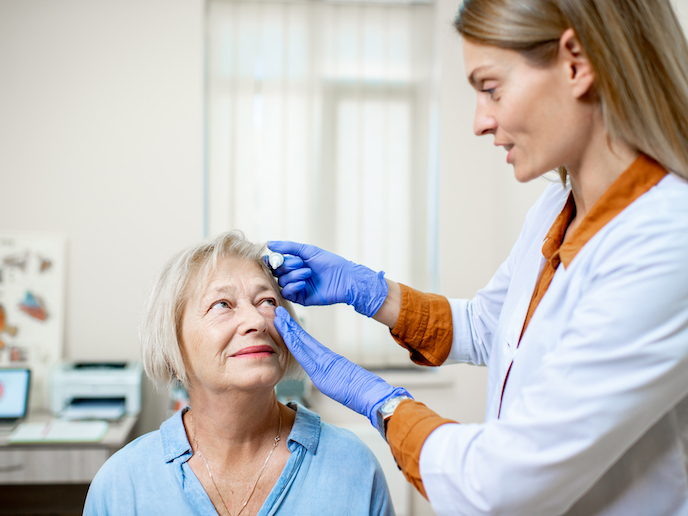
Eye drops specially formulated to prevent and treat retinal damage
Among working-aged adults, diabetic retinopathy (DR) is the main complication of diabetes and the leading cause of blindness. About 110 million people worldwide and 3 % to 4.1 % of Europeans suffer from this disease(opens in new window). Although the early stages of DR usually have no symptoms, when the disease progresses it can lead to retinal neurodegeneration, retinal capillary leakage and blockage. Typical problems include blurred vision, seeing double, difficulty reading, spots or floaters in the field of vision, and loss of peripheral vision. Ophthalmologists can detect the disease very early. However, current treatments, which take the form of injections into the eye or lasers, are quite aggressive and have significant side effects. They tend only to be used in the disease’s advanced stages. From the time the disease is detected until the first symptoms appear, the only thing patients can do is to control glucose levels and blood pressure.
Eye drops targeting retinal neurodegeneration
“With our RetinDR formulation, we are offering a therapeutic solution to these patients for the very first time. Available as eye drops, RetinDR is a topical ophthalmic formulation that effectively prevents the progression of the disease and regresses the symptoms of retinal neurodegeneration caused by diabetes,” says Marta Guerrero, from the EU-funded project RetinDR(opens in new window), and co-founder and CEO of Retinset. The company, a spin-off of www.medicalmix.com (Medical Mix) (website in Spanish), is designing several medicines to treat ophthalmic-related pathologies due to neurovascular degeneration, like glaucoma.
A breakthrough drug delivery formulation
Guerrero, an associate professor of pharmacy and pharmaceutical technology at the University of Barcelona(opens in new window), adds: “A key to our project’s success was increasing the drug’s capability to penetrate the eye and reach the retina, the innermost coat of the back of the eyeball, and responsible for vision. We have developed a formulation platform that enables the eye drops to pass through physiological barriers and to reach the retina.” She notes that the project helped the team to achieve its strategic objectives. These included all the preclinical studies(opens in new window) required before testing the formulation in humans, and starting the phase 1 clinical trial to test the safety in humans. “Today we have the clinical information from the first patients to be administered RetinDR. The results are very promising! We obtained a robust, safe, effective and stable formulation, with high quality as well as an optimal manufacturing process.” Under the project, a market study and business plan allowed the team to better grasp its potential market for the eye drops, including future competitors and technologies. The project also boosted the company’s visibility – thanks to a website launch and attendance at one of Europe’s biggest life science partnering events BIO-Europe Fall(opens in new window) in 2019. “The feedback we received was key to knowing first-hand what investors and the scientific community need to know about RetinDR,” concludes Guerrero. “By delivering drugs to the retina in an eye drop formulation, we are bringing to the market a unique solution for those who suffer from diabetic retinopathy, which has no treatment today for the disease’s first stages.”