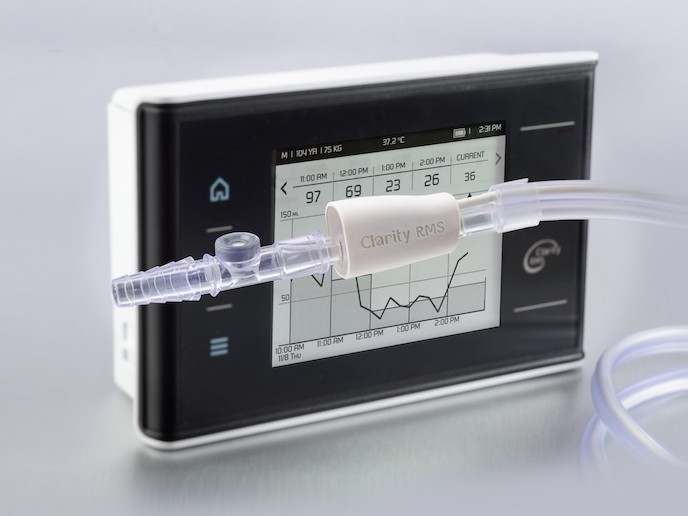
Dataflow: a mobile urine monitoring system for early detection of kidney injury
Every year, 1.7 million patients worldwide die from a rapid loss of kidney function(opens in new window), known as acute kidney injury (AKI). The condition is strongly associated with extended hospital stays, with AKI developing in more than half of patients in intensive care units, where it carries a five-fold increase in mortality. Early detection of AKI and rapid intervention can save lives and reduce the length of hospital stays. The EU-supported Clarity project aims to make this happen through the development of an innovative sensor attached to a Foley catheter, which automatically and continuously monitors urine flow and notifies medical staff when problems arise.
Fluid dynamics
“A lot of companies have tried and not succeeded, because urine is not easy to measure,” says Jean-Paul Benhamou, Clarity project coordinator. “The flow is anything from almost nothing to 300 ml an hour, and the viscosity changes depending on what you eat and your treatment plan.” Typically, data on urine output is collected manually by staff, who inspect a collection bag and record their measurements every few hours. Not only is this labour-intensive, it is also not consistent or reliable enough, adds Benhamou: “In addition, ‘non-contact’ data transmission by an electronic system can reduce the risk of contamination.”
Wireless data
Clarity RMS , developed by Israeli med-tech company RenalSense(opens in new window), is focused on critical care units, and continuously monitors urine flow using temperature exchange. It provides healthcare staff with real-time patient data and notifications of fluctuations via a bedside console and the hospital Electronic Medical Records system. “With the Clarity Stream’s wireless data transmission from the Foley catheter, medical staff will be able to monitor their patients from anywhere in the building. In addition, patients at home will be followed via a mobile app,” explains Benhamou, RenalSense VP sales & marketing, global. “The end goal is precision medicine.” As well as preventing AKI, Benhamou says the device can prove invaluable with monitoring fluid balance, both in hospitals and in situations where kidney function needs to be characterised, such as care homes. Following a successful trial at Herzog Hospital in Jerusalem(opens in new window), RenalSense is launching another trial at a hospital in France this year.
Shrink to fit
To reach its potential, notes Benhamou, the device needs to be miniaturised further so that it can fit entirely within a Foley catheter. “The sensor in our existing product, a few centimetres in size, will be reduced to a few millimetres.” Through EU support, RenalSense was able to carry out a feasibility assessment of the Clarity Stream device, further develop its market strategy, and lay out plans for future technical developments. “It was a proof of concept of the new product line,” adds Benhamou. “After our success with the first-generation Clarity device, we are confident about moving forward with the second generation. The design blueprints are in place, and we are seeking additional capital to implement them.”