Fresh light on a genetic eye disorder
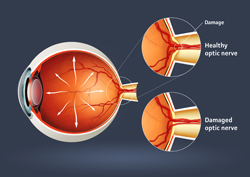
A team of scientists working under the EU-funded project 'Cellular and molecular mechanisms of the light response in photoreceptor cells of the mammalian retina' (Rodcell) is working to discover intracellular signalling events taking place in rod and cone photoreceptor cells, and uses a mouse model to study how genetic flaws lead to retinal degeneration. The primary aim is to describe what happens in the photoreceptor cells of the mammalian retina in response to light. This will help characterise their relevance for cell function and viability. Researchers are combining mouse genetics with morphological, biochemical and electrophysiological analysis. They have already generated a mouse model that will represent a sub-group of mutations in the relevant genes. And the project partners believe this could be a prototype for a wider range of retinal disorders caused by autosomal dominant mutations. This means that the gene carrying a mutation is located on one of the autosomes - chromosome pairs 1 through 22. In terms of inheritance, 'dominant' means that having a mutation in just one of the two copies of a particular gene is all it takes for a person to have a trait, such as the retinal disorder. Initial results indicate that harmful genes and mutations lead to toxicity by affecting the ability of the protein to be properly delivered to the rod outer segment. The protein is not allowed to 'fold' properly and so its accumulation is likely to be responsible for causing proteotoxicity and a stress response leading to cell death. The Rodcell project is now working to characterise the 'stress response' in this transgenic pathway.