The timing of cell fate in the developing embryo
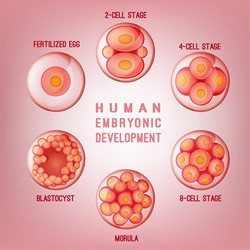
After fertilisation, different cell types can be identified in the 32-64 cell embryo. However, there is still a hot debate on whether there are developmental differences at play well before induction of pluripotency in the inner cell mass (ICM) that becomes the embryo or the trophectoderm (TE) that gives rise to the placenta. During previous research, the SIEAVD team had established an assay to analyse the progression of pre-implantation development. The fluorescence decay after photoactivation (FDAP) assay enabled the quantitative analysis of a key transcription factor, Oct4 that controls pre-implantation progress in different stages of the mouse embryo. The EU funded SIEAVD project systematically used advanced imaging tools to detect the very earliest signs of developmental differences to predict cell lineage patterning events. Particularly interesting, the researchers could predict pluripotency in the embryo as early as the 4-cell stage. Cells with slow Oct4 kinetics and a high immobile fraction were likely to become part of the ICM whereas most cells with fast Oct4 kinetics were destined to become the TE. To further hone in on the protein dynamics at play, the researchers used in vivo imaging to visualise green-to-red photoconvertible fluorescent proteins (pcFPs). Not only did this make high-throughput investigation more available but the green and the red protein populations can be tracked in parallel. SIEAVD also harnessed the decreased phototoxicity made possible using the primed conversion method. By refining the process, the team opened up the possibility for non-invasive high-contrast selection of targeted cells and proteins. By uncovering the molecular mechanism of primed conversion, the researchers created primed convertible variants of most known green-to-red pcFPs as well as several photoconvertible sensors and activity modulators. This dramatically increases the number of trackable proteins from one to all known anthozoan pcFPs. The project has used refined prime conversion as a powerful tool to look at the emergence of asymmetry in the mammalian embryo. Having established primed convertible fast volumetric imaging as a superior imaging methodology, the path is now open to follow the development of all mouse blastomeres with an unprecedented level of precision in time and space.