Non-invasive test for sinus conditions
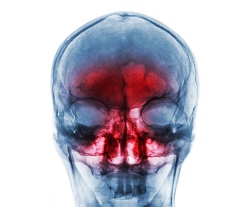
Sinusitis and its causes can be very difficult to diagnose objectively. Methods include nasal endoscopy, imaging using computer tomography (CT) or MRI to detect deep-seated inflammation, nasal cultures and possibly allergy tests if an allergic reaction is suspected. As the causes of sinusitis are many and varied, the patient may have to present for more than one of these procedures. GPX Medical AB, specialists in developing medical technology for non-invasive laser spectroscopy gas concentration measurements in air-filled areas of the body such as the sinuses, developed the concept of a handheld medical device to provide gas volume assessment as an aid for diagnostics of sinusitis in 2012. The technology is based on spectroscopic methods developed by scientists at Lund University, Sweden. Since its development, the company has extended its application to gas measurements in the lungs of prenatal babies. Business plan development With funding from the EU, under the project SINUSLIGHT, GPX Medical reviewed their current financial state and external environment. “In particular,” states Dr Märta Lewander Xu, CEO of GPX Medical, “we mapped the value proposition for end users, analysed markets and competition, and defined the requirements for clinical validation.” The review also focused on the development of a business strategy in accordance with GPX Medical’s vision, values, and long-term development and revenue goals. “We have established how different commercialisation scenarios and potential out-licensing deals influence economics of the company in the long run,” explains Dr Lewander Xu. Antibiotics overuse a significant issue The GPX Medical technology, named GASMAS (gas in scattering media absorption spectroscopy), is unique in that it can, non-intrusively, measure gases situated in cavities enclosed in highly scattering and absorbing surrounding tissue. The technique is based on transmitting low-power laser light with the sensor positioned on the surface of the object. This makes it possible to measure the concentration of gases in body cavities non-invasively. Measurement using the eye-safe spectroscopic laser technique takes a few seconds and the result is immediately displayed. The device provides information on the open/air-filled sinus cavity size, the oxygen concentration in the cavity and therefore its ventilation status. “The improved diagnostics provided by the device can provide an objective test and improve the care and reduce the unnecessary prescribing of antibiotics,” emphasises Dr Sara Bergsten, project leader with SINUSLIGHT. “The technology has already been clinically evaluated in a research study of 40 sinus cases with good success in correlation with the golden standard CT,” she adds. Company vision focused with a view to commercialisation “This renewed review was very much needed and confirmed some of the conclusions from 2012 in terms of the challenges on the technical side and acceptance from the general practitioners,” says Dr Lewander Xu. Results of the SINUSLIGHT project have reinforced that there is a very clear need for the GPX Medical device in terms of global health. However, it requires evidence-based documentation to convince the medical doctors to use the technology. Market analysis needs to be performed at a high level, approaching organisations like the World Health Organization as well as individual nations’ policymakers. This is needed to discuss the value and reimbursements for a technology that could lower the number of deaths related to antibiotic resistance. Dr Bergsten sums up the massive implications brought about by the SINUSLIGHT review: “We see a great business potential in both applications, sinuses and lungs, and are now starting to execute the steps needed for taking these technologies to the market.”