Surgical innovation could make operations safer
As nearly 30 % of diseases are surgically treatable, it comes as no surprise that there are over 2 million surgeries conducted every year in developed countries. While many of these procedures are categorised as minor and non-life threatening, over 10 % involve the dissection of the pathological tissue from the surrounding healthy tissues. Even though such dissection is a critical procedure, surgeons continue to use outdated tools and methods. “Despite the importance of selective dissection in surgeries, current tools still rely on either tearing force or non-selective cutting,” says Benoît Verjans, CEO of AuXin Surgery(opens in new window) and coordinator of the EU-supported CADISS project. “All too often, this results in critical organs, such as the nerves and muscles, being damaged or in an incomplete tumour withdrawal.” To change this, AuXin Surgery has developed the CADISS system(opens in new window), an innovative new technology for separation surgeries. “CADISS decreases the time patients spend in operating rooms and reduces the risks of complications,” adds Verjans. Thanks to EU funding, the company has conducted an in-depth feasibility study of its technology – an important first step towards bringing its game-changing device to market.
Significant benefits to patients
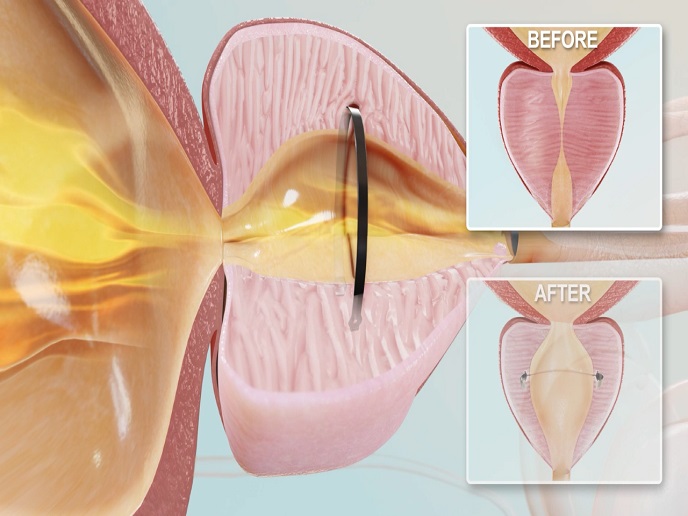
The CADISS system is unique in that it uses a combination of mechanical surgical instruments and the topical deposition of a drug. Together, this allows for the separation of pathological tissues without the need for cutting instruments. “With the drug weakening fibrosis(opens in new window), the instrument is able to easily separate the two organs,” explains Verjans. “This makes CADISS especially useful when there is a significant risk of a major problem if one of the tissues is damaged.” With the EU funding, the company was able to confirm the system’s technical feasibility and commercial profitability. For example, tests showed that CADISS reduces the risk of damaging the membrane protecting the spinal cord from 30 % to just 10 %. The research team also observed a 20 to 30 % reduction in surgery times. “Most importantly, we proved the novelty of our innovation and the significant benefits it will bring to patients and the healthcare sector,” notes Verjans. “These benefits include fewer side-effects, fewer tumour relapses, and shorter surgery times.”
Improving quality of life
Despite several early setbacks with manufacturing and testing the device, AuXin Surgery is well on its way to commercialising its CADISS system. “We succeeded in fully developing the device, testing it in clinical trials, and registering it for approval by both DEKRA(opens in new window), the medical device certification authority, and FAMHP(opens in new window), the pharmaceutical authority,” adds Verjans. “As a result, several patients have already experienced a radical improvement in quality of life thanks to fewer surgical side-effects and relapses.” The company is currently working on marketing CADISS worldwide, having recently signed distribution contracts in 17 countries. They are also seeking a second round of funding to further develop the device and support its market launch.