Targeted approach offers hope to ovarian cancer patients
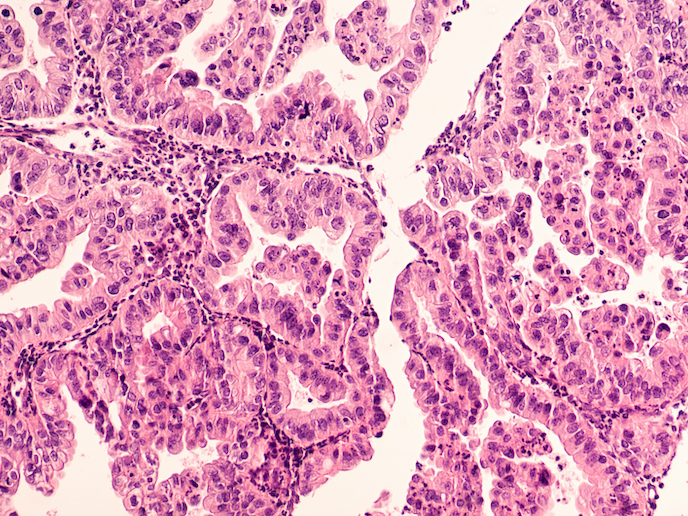
Ovarian cancer, which kills more than 40 000 women in Europe every year, has a very low survival rate. Only about a third of diagnosed patients will live for another 5 years. “A key reason for this is that this cancer is usually detected very late,” explains HERCULES(opens in new window) project coordinator Sampsa Hautaniemi, professor of Systems Biology at the University of Helsinki(opens in new window), Finland. “It is often called the ‘silent killer’ because it has few symptoms, and when discovered, has already spread to the abdomen. At this stage, the cancer is very difficult to treat.” While surgery at this stage is critically important for removing tumours, identifying all cancerous cells from such a large area is practically impossible. Surgery is therefore typically followed by courses of chemotherapy, which can help extend patient lifespans. After about 2 years however, patients often stop responding to treatment, and the cancer returns.
Tackling drug resistance
The focus of the HERCULES project was therefore to improve outcomes for ovarian cancer patients, by examining why cancer cells become unresponsive to treatments. “Our aim was to identify the specific mechanisms that cause tumours to become resistant to chemotherapies,” adds Hautaniemi. DNA and RNA sequencing(opens in new window) were applied to cell samples from over 180 ovarian cancer patients, and an unprecedented amount of information about tumour cell populations gathered. Data from these experiments were used to establish computational models, to help predict the most effective drug combinations to kill cancer cells. “What we found was that there are typically one or two dominant resistant mechanisms for chemotherapy in cancer cells,” says Hautaniemi. “With this knowledge, we were able to test if existing drugs could be combined with chemotherapies to tackle this resistance, and give patients longer to live.”
Improved life expectancy
“One of the things I am proudest of is that we showed a proof of concept for the personalised medicine approach which HERCULES was based on,” remarks Hautaniemi. “In one relapsed, chemoresistant patient for example, we identified the dominant resistant mechanism, identified the right drug for her, and included this in her therapy. The response to this combination treatment was excellent.” The project also developed new drug screening techniques, as well as methods to increase the culture of organoids(opens in new window). Cell lines from patient samples were also developed to examine responses to specific anti-cancer drugs in the wet lab. Based on all this work, a prototype for predicting the best drug combinations for individual patients was developed. “All this will help scientists involved in discovering therapy targets based on patient samples,” says Hautaniemi. “The fact that we made so many practical advances in what was a research project is something that I am very happy about. Such work is vital, because I think ovarian cancer remains somewhat understudied.” Data generated in the HERCULES project will be available in the European Genome Archive. The advances made during the HERCULES project are now being applied in a new EU project called DECIDER. “Our aim here is to have more clinical impact,” explains Hautaniemi. “We will continue to collect patient samples, but our focus will be on detecting patients who might soon relapse. The window for action is quite narrow, as patients without effective treatment may deteriorate rapidly. Therefore, we have to be faster to collect and analyse the data. Without the HERCULES project this would not be possible.”