Treating intractable paediatric epilepsy
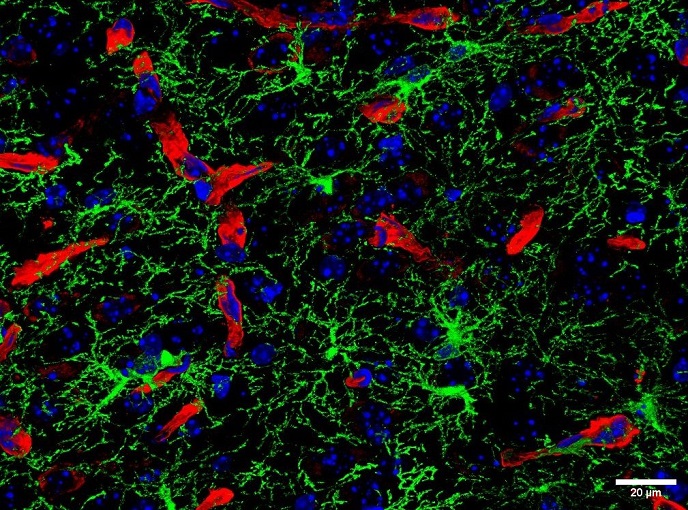
The DESIRE (Development and epilepsy - strategies for innovative research to improve diagnosis, prevention and treatment in children with difficult to treat epilepsy) project is focusing on epileptogenic developmental disorders (EDDs) which are early onset paediatric epilepsies related to developmental brain processes. The work is particularly significant as EDD is poorly understood and affected children often develop early drug resistance and cognitive decline. DESIRE aims to elucidate the genetic and epigenetic causes and pathogenic mechanisms of EED to identify new biomarkers for diagnosis and prognosis, and to discover and improve therapies. A clinical trial is underway to determine if treating children with ketogenic diet prior to surgery for focal cortical dysplasia (malformation of the cortex) can improve prognosis. So far, genomic research to find underlying causes has involved analysis of over 300 families with epilepsies. State of the art gene sequencing was performed as well as expression analysis and epigenomic profiling. New disease-causing genes and genetic variations were identified that could be linked to developmental malformations of the brain. High-density autosomal maps of methylation sites allowed to identify novel epigenetic gene imprints. Use of sophisticated animal models pinpointed migration of neurons as an epileptogenic mechanism. Neurophysiological biomarkers were explored using non-invasive diagnostic prototype tools that can analyse high frequency oscillations indicative of seizure onsets. Hardware and software needed for this novel diagnostic approach are pushed forward by the company partners in the project. Since to date, pharma industry has treated the complex range of epilepsies as one disorder thereby largely ignoring diverse causes and diverse treatment needs of paediatric patients, DESIRE aims to explore at pre-clinical stage a battery of novel treatments, spanning from genetically modifying neurons to express new metabolites, and grafting inhibitory or silenced neurons, to employing nanoparticle carriers to drive therapeutic protein compounds across the Blood-Brain-Barrier to their brain targets, leading to attenuating or preventing seizure onset. DESIRE adds a personalised therapy approach to epilepsy treatment in children, using the currently most advanced technologies. The project is based on a strong collaboration between the multidisciplinary research teams at hospitals and small and medium enterprises, to ensure the ambitious but very relevant objectives are met.