Copper is the metal culprit in Alzheimer’s disease
Alzheimer’s disease (AD) was first officially described in 1906 and the classification ‘Alzheimer’s disease’ was included in a psychiatry textbook a few years later. Despite more than a hundred years of clinical data and research since then, many questions remain. The aLzINK project, funded by the EU’s European Research Council (ERC), has answered an important one, pointing the way to an important target for drug candidates that could prevent the development of this devastating degenerative neurological disease.
Not all metals are created equal
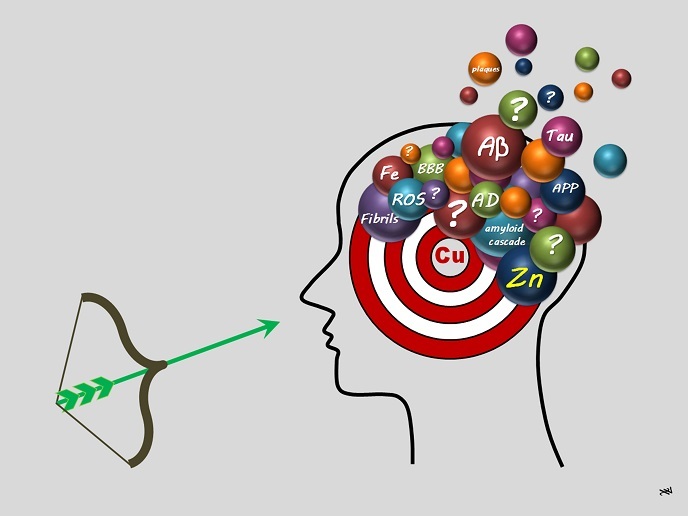
For the last 30 years, the amyloid cascade hypothesis(opens in new window) has been a key focus of AD research. It proposes that the aggregation of amyloid-β(opens in new window) (Aβ) peptides (where peptides are two or more amino acids linked in a chain) is an early and the first key event. The second is the formation of reactive oxygen species (ROS)(opens in new window) related to oxidative stress(opens in new window). At the final stage, self-assembled peptides are gathered in so-called senile plaques, a hallmark of the disease. Metal homeostasis (maintaining the appropriate metal balance required for physiological functioning), particularly of copper (Cu) and zinc (Zn), has been linked to all three steps. ERC grantee Christelle Hureau-Sabater explains: “Cu can be found in two oxidation states, Cu+ and Cu2+. While Cu can trigger ROS formation Zn is inert and cannot. Both ions can link to Aβ peptides, but in different ways, thus modulating Aβ peptide aggregation differently. One therapeutic strategy relies on removing metal ions bound to Aβ peptides. We hypothesised that Cu should be selectively removed, which makes the design of therapeutic molecules/drug candidates (called ligands or chelators) more difficult.” The aLzINK project set out to develop guidelines to do so.
Overcoming challenges, hitting the therapeutic ‘bull’s eye’
The Aβ peptide is a short, intrinsically disordered protein of about 40 amino acids that is derived from a longer membrane protein. Its flexibility and lack of a preformed 3D structure make it difficult to identify where Cu and Zn bind to the peptide and how. According to Hureau-Sabater: “We overcame this issue by using many complementary techniques including sophisticated X-ray absorption spectroscopy(opens in new window) or XAS.” XAS is widely used to determine the local geometric and/or electronic structure of matter relying on synchrotron radiation for intense and tuneable X-ray beams. Thanks to innovative protocols and collaboration with other groups, Hureau-Sabater’s team overcame challenges, successfully meeting its original objectives. “We have precisely shown why AD drug candidates should specifically target Cu. We tested several molecules in solutions and some in cells with promising results.” This provides a starting point for future studies of the therapeutic potential of Cu chelators (sequestering agents) and related compounds. Along the way, the researchers also highlighted the utility of inorganic prodrugs instead of the purely organic parent ligand and discovered the importance of exchange rates between the ligands Aβ, Cu and Zn and of targeting the Cu+ state of copper. Hureau-Sabater concludes: “Many therapeutic approaches to AD have failed, including those targeting metal ions. In aLzINK, we have demonstrated that many criteria must be considered, and we have elucidated important features regarding those criteria.” aLzINK has opened a new door on more precisely targeted therapies for AD that could enhance benefits while minimising unnecessary side effects.