Reopening the gateway from the heart to the body
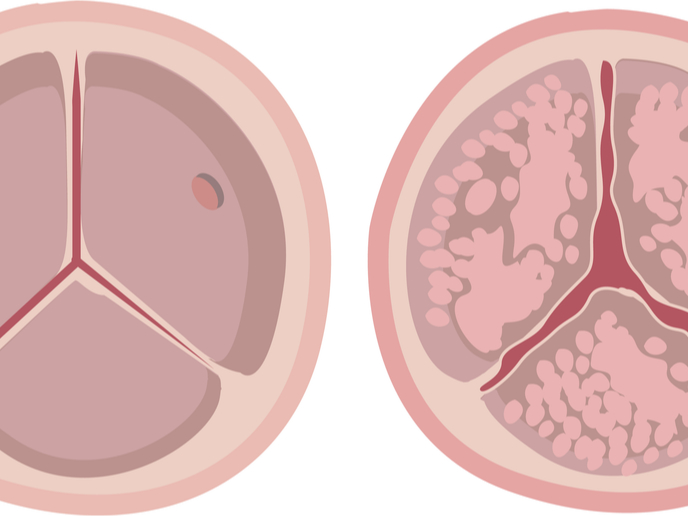
Two million people in the Western world are in danger of dying suddenly from aortic stenosis(opens in new window) (AS). Stenosis means narrowing: calcium deposits in the aortic valve cause stiffening and rob the valve of flexibility, resulting in poor blood flow. The standard treatment is valve replacement. This is done either through risky open-heart surgery, or through endovascular transcatheter aortic valve replacement, delivered via a leg vein – also with serious complications. However, valve replacement is only available to chronically sick patients. In fact, three-quarters of sufferers go untreated, and are in danger of developing severe AS. “This represents a huge unmet medical and economical need,” says Benjamin Bertrand, coordinator of the EU-funded project OUTLIVE and CEO of the host company French medtech, CARDIAWAVE SA(opens in new window).
Gamechanger for all AS patients
CARDIAWAVE’s alternative therapy, Valvosoft, repairs the aortic valve and doesn’t involve replacement. Importantly, unlike current medical solutions, this is a non-invasive treatment with low risks, complications and costs. So how does Valvosoft work? “The device delivers targeted, high-intensity ultrasound waves which break down the brittle calcium and soften the tissue,” explains Bertrand. This gives the aortic valve a new lease of life by repairing and restoring its aortic valve function, allowing blood to flow unhindered to the body.
Re-engineered to improve accuracy and refinement for safety
After the first set of clinical trials, CARDIAWAVE improved the safety, performance, usability and maintenance of the Valvosoft device. They reengineered almost all hardware subsystems and software modules together with the graphical user interface. These measures enhanced image quality and therapy-targeting accuracy as well as increasing the power the device can deliver. For extra data on safety, an MRI procedure was implemented for patients, before and after the therapy. Designed to demonstrate the absence of stroke risk, this was unfortunately suspended in the wake of the COVID crisis. However, none of the MRIs collected to date detected any abnormalities. These results were published in the high impact journal Circulation(opens in new window) in 2021.
And now to market
The objective of the OUTLIVE project was to bring to market the Valvosoft device, transforming a very promising clinical prototype into an industrial approved medical tool. “The clinical investigation and the technical development of the Valvosoft device and its manufacturing process represent the major part of the work carried out within OUTLIVE,” says Bertrand. A refined go-to market study resulted in a new, detailed financial, marketing and commercialisation plan to establish revenue streams and pricing. Agreements have been secured with key suppliers and subcontractors that manufacture essential parts of the device. A key objective of the OUTLIVE project was to obtain CE marking. In readiness, the new version of the device will be used in subsequent clinical investigations; researchers expect to secure the conformity marking by 2023, after the new round of clinical trials. Mobile and non-invasive ultrasound therapies are gaining momentum generally. Principal investigator on Valvosoft, Professor Emmanuel Messas, sums up: “We have every confidence in the success of the next stages of its clinical development, which we expect to accelerate with our next fundraising planned for 2022. Valvosoft will become a new gold standard in AS treatment.”