Intercepting molecular interactions
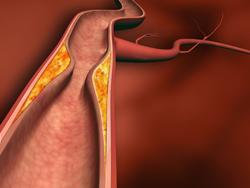
The make-up of low-density lipoprotein (LDL) particles can be altered through enzymatic degradation, aggregation and oxidation. Now, research indicates that complement may play an important part in this modification process. An intricate system of enzymes and regulators, complement plays a major role in helping an organism defend itself against pathogens. The multimolecular protease C1 triggers the complement pathway and participates in other related biological functions thanks to the binding properties of its C1q subunit. Researchers have previously studied the ability of enzymatically modified LDLs (E-LDL) to activate C1 as they can be recognised by C1q — suggesting that complement activation by E-LDLs may heavily influence the development of atherosclerosis. The 'Complement and atherosclerosis: molecular mechanism of C1 activation by enzymatically modified low-density lipoproteins' (Complement and LDL) project set out to characterise the processes leading to C1q recognition of E-LDLs. This was to be achieved by identifying the molecules that the subunit recognises and the mechanisms leading to it, as well as analysing the processes underlying C1 activation by E-LDLs. The EU-funded project took a molecular and structural level approach employing protein biochemistry methods, functional studies, surface plasmon resonance spectroscopy and X-ray crystallography. Study results all indicate that C1 binding to E-LDL involves C1q recognition of free fatty acids generated by cholesterol esterase (CEase) treatment. On the strength of such investigations, researchers hypothesise that C1q may work to prevent or control programmed cell death (apoptosis) by binding to E-LDL particles. As such, the potential to sense and clear apoptotic cells, as well as regulate fatty acid-induced apoptosis is an important discovery in understanding more about the atherogenesis process. Complement and LDL project results could therefore contribute to future efforts to develop novel therapies against atherosclerosis. Mapping the interaction between E-LDL and C1q may shed light on how to design and synthesise molecules capable of modulating this interaction, and thus control or prevent complement activation.