MicroRNA deregulation in CRC patients now exposed
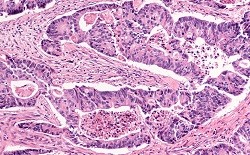
The link between MicroRNA deregulation and colorectal cancer (CRC) progression needs no further evidence. Yet, when he applied for EU funding in 2013, Dr Valeri could only note how researchers were still not entirely sure of why and how this deregulation occurred: ‘Despite several groups identifying a link between microRNA deregulation and bowel cancer, no one had put microRNA deregulation in the context of recurrent abnormalities observed in early initiation and progression of bowel cancer,’ he recalls. To close this gap, the leader of the Gastrointestinal Cancer Biology and Genomics Team within the ICR’s Centre for Molecular Pathology has spent the past four years addressing two important questions. First, he assessed how genetic and epigenetic events contribute to controlling microRNA genes and cancer phenotype. Then, he looked into how synthetic lethal interaction between microRNA genes aberration and kinase genes can help define novel targets for therapy. The research mostly consisted in three steps. Dr Valeri and his team began by screening cases of primary and metastatic colorectal cancers using publically available databases and retrospective series collected from different institutions in Europe and the US. Then, they performed high-throughput siRNA and drug screenings to define vulnerabilities and mechanisms of resistance associated with microRNA deregulation. Finally, they validated hits from their screenings in vitro and in vivo by using colorectal cancer material from over 2500 patients. ‘All in all, we tested microRNA deregulation in genetically-engineered mouse models, in their derivative organoids, in human and mouse cell lines harbouring specific mutations in oncogenes, and in tumour suppressor genes such as APC, KRAS, TP53, PIK3CA and SRC,’ Dr Valeri explains. ‘We also investigated microRNA expression in tissues of bowel cancer patients enrolled in clinical trials and, in order to study the contribution of specific microRNAs in relation to an abnormal oncogene, we modulated the expression of the microRNA and could observe changes in cancer behaviour like proliferation, cell death, invasion, migration and metastasis.’ Towards more effective, target treatments With this research, the team was able not only to identify key microRNAs involved in the initiation and progression of bowel cancer, but also to providence evidence that these might act as downstream effectors of oncogenes and tumour suppressor genes frequently mutated in bowel cancer. ‘Targeting and blocking these microRNAs might result in the development of novel therapeutics,’ Dr Valeri enthuses. Dr Valeri also showed that mutations in microRNAs genes might predict outcome and response to treatment in patients with locally advanced rectal cancer. In other words, the detection of these abnormalities might result in better patients’ selection and personalised medicine. When asked how soon such medicines could be commercialised, Dr Valeri prefers to remain cautious. Whilst MicroRNA inhibitors are already being tested in clinical trials to treat several medical conditions including chronic infections and lung cancers, such approaches will require early phase clinical trial validation over the next few years. In the meantime, Dr Valeri intends to pursue his research. ‘Through the award of further research funding, I will continue to investigate the contribution of microRNAs in colorectal and gastroesophageal cancers. I am confident that this approach will help us identify novel therapeutic targets and potential biomarkers useful in patient’s selection and precision medicine,’ he concludes.