Accelerating innovation – and chest trauma treatment
Getting a medical device from concept to clinic is a long, complex and heavily regulated process – but one that can be streamlined with the support of EU funding. Take Vigor Medical Technologies(opens in new window) and its EU-funded C-LANT Drainage Port(opens in new window) project as a case in point. This medical device company received EUR 2.5 million in funding from the EIC Accelerator(opens in new window), which it used to help accelerate the development, testing and commercialisation of its C-Lant drainage port.
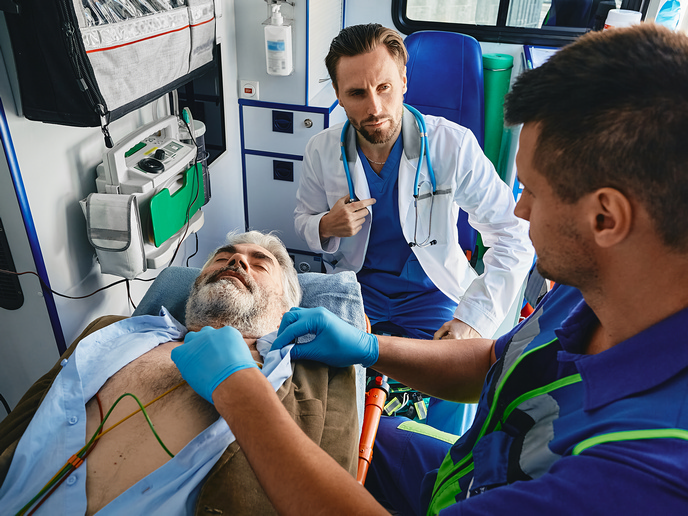
Innovative medical device increases chance of surviving chest trauma injury
Chest trauma is the leading cause of accidental death globally. But if promptly treated – ideally within an hour of injury – patients can have an 80 % chance of survival. “Unfortunately, because most of the available treatment options require a high-risk chest tube procedure to drain fluids and air from the thoracic cavity, they can only be used by highly qualified professionals,” says Vigor Medical Technologies CEO Irina Kavounovski. “Even then, these procedures can have a 40 % complication rate.” The C-Lant drainage port, on the other hand, is safe and easy to use. In fact, paramedics in the field, the ambulance or the hospital can easily use the port to drain the chest cavity within the critical timeframe, reducing procedure time from over 20 minutes to just a minute and a half. “This revolutionary device enables emergency first responders to immediately administer crucial treatment to victims, significantly improving the chances of survival and positive outcomes for those in critical conditions,” adds Kavounovski.
Towards operational readiness and future market entry
Thanks to the support of EU funding, the life-saving C-Lant drainage port has already been approved for clinical use in the United States and is on the cusp of approval in Europe. In addition to helping complete the development of the device, the funding allowed Vigor Medical Technologies to establish a coherent financial, legal and administrative framework fully aligned with the device’s commercialisation objectives. “The project budget was deployed in line with the approved cost categories to secure high quality corporate legal services, to structure relationships with key suppliers and expert advisors, and to build the necessary European corporate footprint – all of which are critical to ensuring that all expenditures directly support operational readiness and future market entry,” notes Kavounovski.
Tests confirm C-Lant port is safe, fast and effective
The EU funding was also used to confirm the C-Lant’s benefits. This process involved conducting over 30 verification and validation tests, which ultimately showed that the device was not only safe, but also fast and effective. “The C-Lant can be inserted in seconds, resulting in a 20-fold reduction in procedure time and addressing 60 % of the polytrauma mortality currently associated with treating chest trauma caused by car accidents and falls,” explains Kavounovski. These tests led to the device receiving ISO 13485 and FDA certifications, paving the way for its use in both the EU and American markets.
Eyeing European and global markets
With a subsidiary established in Czechia, Vigor Medical Technologies is primed to begin mass-producing its C-Lant port. “Thanks to the EU’s comprehensive support, we are now set up to pursue the necessary clinical, regulatory and commercial scaling of the C-Lant drainage port in both European and global markets,” concludes Kavounovski.